Health communication
CONTENTS
Amebic dysentery
1. General Overview
Amebiasis is an invasive infection caused by Entamoeba histolytica (E. histolytica), a parasite that is widely distributed worldwide. Approximately 90% of infections are asymptomatic, whereas the remaining 10% present with a broad clinical spectrum, ranging from dysentery syndrome to hepatic abscess and other extraintestinal manifestations.
Amebic dysentery is the typical intestinal form of the disease, characterized by bloody mucoid diarrhea, abdominal pain, and tenesmus. Fulminant amebic colitis is rare but may rapidly become fatal. Severe complications include colonic perforation, colonic ulceration, ameboma formation, and chronic parasitic carriage.
The disease is transmitted mainly through the gastrointestinal route in areas with poor sanitary conditions, where food and water are easily contaminated with human feces. In endemic regions, amebic dysentery may account for up to 40% of diarrheal cases.
2. Causative Agent
E. histolytica is an anaerobic unicellular protozoan belonging to the genus Entamoeba in the family Entamoebidae. Several Entamoeba species inhabit the human intestinal tract, including E. histolytica, E. dispar, and E. moshkovskii; however, only E. histolytica has been definitively established as pathogenic.
The life cycle of E. histolytica consists of four stages: trophozoite, precyst, cyst, and metacyst. The cyst is the infective form, resistant to gastric acid and capable of prolonged survival in the environment. After reaching the small intestine, the cyst wall is digested, releasing four nuclei; each nucleus divides to form eight immature trophozoites. These migrate to the cecum and colon, where they mature and reproduce asexually. Some trophozoites subsequently encyst and are excreted in the stool, thereby continuing community transmission.
Trophozoites generally do not survive outside the host and are destroyed by gastric juice if ingested. In some cases, however, they invade the intestinal mucosa, causing amebic dysentery, or spread hematogenously to the liver, lungs, brain, and other organs, resulting in extraintestinal disease.
Amebic cysts are highly resistant: they can survive for several days in feces, at least 8 days in soil at 34–38°C, up to 1 month at 10°C, and remain viable in cold water, seawater, or wastewater. Cysts are inactivated by iodine at 200 ppm, acetic acid at 5–10%, temperatures above 68°C, or by boiling drinking water for 10 minutes.
3. Epidemiology
3.1. Source of infection
Humans are the principal reservoir of infection, particularly chronic cyst carriers.
3.2. Mode of transmission
The disease is transmitted through the fecal–oral route via food or drinking water contaminated with feces from individuals infected with intestinal amebiasis.
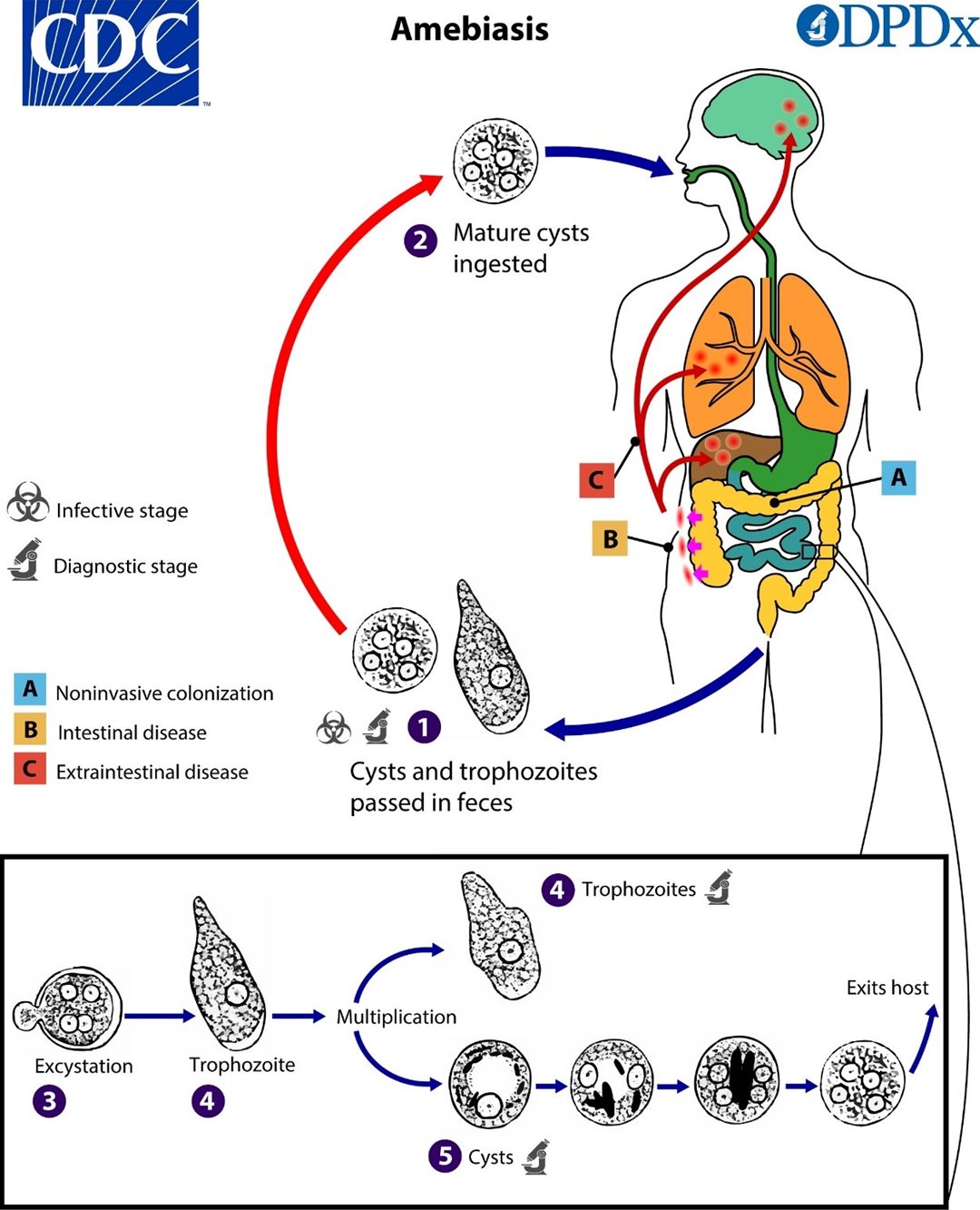
Figure 1. Life cycle of E. histolytica
In which:
- Cysts and trophozoites are passed in the stool
- Humans ingest mature cysts
- Cysts reach the small intestine and release trophozoites
- Trophozoites develop and multiply in the intestinal lumen
- Trophozoites differentiate into cysts → passed in the stool, continuing the cycle
A. Noninvasive infection: amebae remain confined to the intestinal lumen
B. Intestinal disease: amebae invade the colonic mucosa → causing amebic dysentery with bloody mucoid stools
C. Extraintestinal disease: amebae spread hematogenously to the liver, lungs, brain, and other organs → causing amebic liver abscess or other organ involvement.
3.3. Epidemiologic characteristics
Amebic dysentery is widely distributed worldwide, particularly in tropical regions and in areas with poor sanitation. The disease is more common in communities where fresh human feces are used in agriculture or where geophagia is practiced. It may occur at any age; however, amebic liver abscess is approximately ten times more common in adults than in children, whereas young children are more likely to develop fulminant colitis.
Each year, an estimated 50 million people worldwide develop amebic dysentery, with approximately 100,000 deaths, mainly due to severe complications of invasive disease, particularly amebic liver abscess.
4. Pathogenesis
E. histolytica is capable of destroying multiple tissue types, most commonly the intestinal mucosa and liver, and less frequently the lungs, brain, bone, or cartilage.
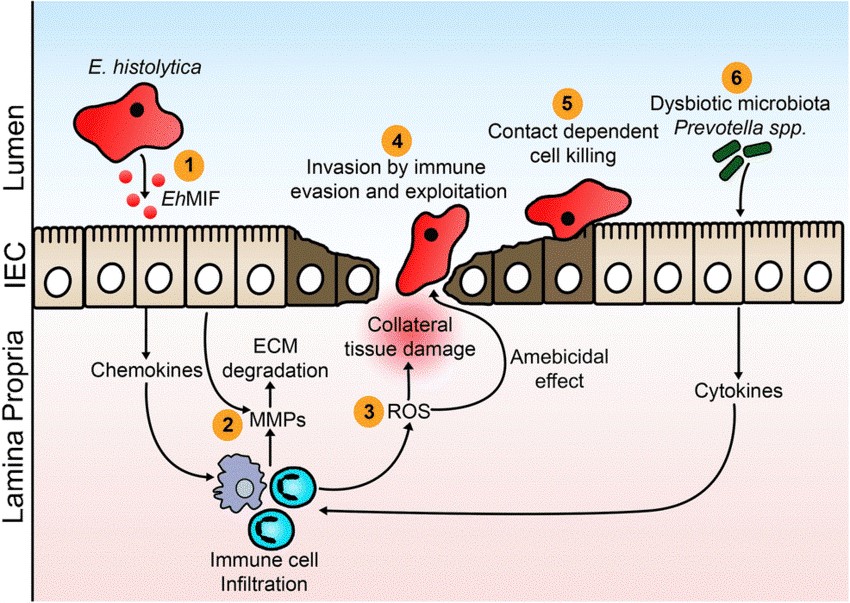
Figure 2. Pathogenic mechanisms of E. histolytica in the intestinal tract
- Macrophage migration inhibitory factor (EhMIF) secreted by E. histolytica promotes mucosal inflammation.
- Inflammation induced by E. histolytica increases the production of matrix metalloproteinases (MMPs), which degrade the extracellular matrix (ECM) of the intestine and facilitate cell migration.
- Infiltrating inflammatory cells generate reactive oxygen species (ROS), which can kill the parasite. These free radicals also contribute to secondary tissue injury during the inflammatory phase.
- E. histolytica invades the intestinal mucosa by evading and exploiting the host immune system.
- E. histolytica kills host cells in a contact-dependent manner.
- Increased abundance of P. copri may increase the risk of colitis.
5. Clinical Manifestations
The incubation period usually ranges from 2 to 4 weeks but may vary from a few days to several months or even years. Approximately 80–90% of infections are asymptomatic. Symptomatic cases may be self-limited or may recur intermittently. The disease usually begins insidiously and progresses gradually, whereas fulminant amebic colitis has a very rapid and severe course.
Typical symptoms
- Abdominal pain, which may be localized or diffuse, commonly involving the cecum, transverse colon, or sigmoid colon
- Diarrhea with mucus and/or blood
- Tenesmus, commonly seen with lesions involving the sigmoid colon and rectum
Systemic manifestations
Systemic symptoms are usually not prominent; weight loss, anorexia, and varying degrees of dehydration may occur depending on the severity of diarrhea, but severe dehydration is uncommon.
Clinical forms of amebic dysentery
- Acute amebic dysentery: bloody mucoid diarrhea, abdominal pain, and tenesmus
- Chronic amebic dysentery: prolonged or recurrent disease, easily confused with chronic colitis or inflammatory bowel disease
- Fulminant amebic colitis: a rare complication (<0.5%) but very severe, with a mortality rate >40% if not treated promptly.
Extraintestinal amebiasis
- Amebic liver abscess: fever, pain in the right upper quadrant or epigastrium, pain on liver percussion, or tenderness over the intercostal spaces
- Less commonly: involvement of the lungs, brain, skin, or other organs.
Important note: amebic dysentery should be excluded before initiating corticosteroids or immunosuppressive therapy for colitis. Corticosteroids may rapidly exacerbate invasive amebiasis and become life-threatening.
6. Complications
6.1. Toxic megacolon
This is a very rare complication of amebic dysentery, usually associated with a poor prognosis and often requiring surgical intervention.
6.2. Fulminant amebic colitis
This is the most severe complication, with a very high mortality rate of approximately 40%. High-risk groups include young children (especially those under 2 years of age), pregnant women, and immunocompromised individuals such as those with malnutrition, corticosteroid therapy, or HIV/AIDS. It has an abrupt onset, with symptoms including:
- High fever
- Severe abdominal pain
- Profuse bloody mucoid diarrhea
- Marked leukocytosis
6.3. Other severe colonic complications
Colonic hemorrhage may occur in severe or extensive disease. Colonic perforation leading to peritonitis is a life-threatening complication, commonly seen in fulminant amebic colitis.
7. Laboratory findings
7.1. Stool microscopy for erythrophagocytic trophozoites
Fresh stool examination is the classical method; however, its sensitivity is low when only a single sample is tested because cyst excretion is intermittent and trophozoites degenerate rapidly. At least three stool specimens collected over 2–3 days are recommended to improve detection.
7.2. Culture
Culture of E. histolytica from stool or rectal biopsy specimens can be performed, but it is technically demanding, time-consuming, and is now rarely used in routine practice.
7.3. Immunologic tests
- Stool antigen detection (ELISA/EIA): specific for E. histolytica, with high sensitivity and specificity
- Serology (antibody detection): highly positive in extraintestinal invasive disease such as amebic liver abscess, but less sensitive in intestinal disease and unable to distinguish past from current infection because antibodies may persist for many years. False-negative results may occur during the first 1–2 weeks of an acute episode.
7.4. Molecular biology
PCR for E. histolytica in stool has variable sensitivity and specificity depending on the sample preparation technique and the target gene used.
7.5. Stool leukocyte examination
Stool examination for polymorphonuclear leukocytes is performed using methylene blue staining. Results are graded according to the number of neutrophils per high-power field (HPF):
- Many: >10 neutrophils/HPF
- Moderate: 5–10 neutrophils/HPF
- Few: <5 neutrophils/HPF
- None: 0 neutrophils/HPF
The higher the neutrophil count in stool, the more suggestive it is of an invasive diarrheal pathogen.
7.6. Hematology
In mild cases, the complete blood count may be normal. In severe or invasive disease, leukocytosis, mild anemia, and an elevated erythrocyte sedimentation rate may be observed.
8. Treatment
8.1. Principles of treatment
- Eradication of tissue-invasive amebae
- Eradication of intraluminal amebae
- Symptomatic treatment
8.2. Specific antiamebic therapy
a) Tissue amebicides
- Metronidazole: 500–750 mg three times daily for 7–10 days in adults; 35–50 mg/kg/day in 3 divided doses in children
- Tinidazole: 2 g/day for 3 days (or 3–5 days)
- Secnidazole: adults 2 g orally as a single dose; children 30 mg/kg.
b) Luminal amebicides
- Metronidazole: 500–750 mg three times daily for 7–10 days in adults; 35–50 mg/kg/day in 3 divided doses in children
- Tinidazole: 2 g/day for 3 days (or 3–5 days)
- Secnidazole: adults 2 g orally as a single dose; children 30 mg/kg
These should be followed immediately, after completion of tissue amebicides, by one of the following luminal agents:
- Paromomycin: 25–30 mg/kg/day in 3 divided doses for 7 days
- Iodoquinol: 650 mg three times daily for 20 days
- Diloxanide: 500 mg three times daily for 10 days.
9. Prevention
9.1. Food and water hygiene
- Continuous education on personal hygiene, especially handwashing with soap before meals and after using the toilet
- Consumption of properly cooked food and boiled water
- Drinking water should be boiled or filtered, because chlorination at conventional concentrations does not kill amebic cysts.
9.2. Water source and waste management
- Strict management of domestic water supplies and wastewater
- Feces and waste from infected patients must be collected and hygienically treated
- Fresh human feces must never be used as fertilizer or in animal husbandry, in order to prevent transmission.
9.3. Management and treatment of patients
- Patients with amebic dysentery should receive specific antiamebic therapy
- Discharge should be considered only after two consecutive stool examinations are negative for amebic cysts
- Clothing and linens contaminated with stool must be properly washed and disinfected.
9.4. Detection and treatment of cyst carriers
- In communal settings such as schools, nurseries, military units, institutional kitchens, and restaurants, food-service workers should undergo stool testing every 6 months to detect amebic cysts
- Identified cyst carriers must receive anti-cyst treatment
- During treatment, temporary reassignment away from food handling should be considered to prevent transmission.
REFERENCES
- Bennett JE, Dolin R, Blaser MJ. Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. Elsevier Health Sciences, 2019.
- Kantor M, Abrantes A, Estevez A, et al. Entamoeba histolytica: Updates in Clinical Manifestation, Pathogenesis, and Vaccine Development. Can J Gastroenterol Hepatol. 2018;2018:4601420.
- Cooney J, Siakavellas SI, Chiodini PL, et al. Recent advances in the diagnosis and management of amoebiasis. Frontline Gastroenterology. 2025;16(1):37–50.
- Lin FH, Chen BC, Chou YC, et al. The Epidemiology of Entamoeba histolytica Infection and Its Associated Risk Factors among Domestic and Imported Patients in Taiwan during the 2011–2020 Period. Medicina. 2022;58(6):820.
- Shane AL, Mody RK, Crump JA, et al. 2017 Infectious Diseases Society of America Clinical Practice Guidelines for the Diagnosis and Management of Infectious Diarrhea. Clin Infect Dis. 2017;65(12):e45–e80.
- Shirley DT, Farr L, Watanabe K, Moonah S. A Review of the Global Burden, New Diagnostics, and Current Therapeutics for Amebiasis. Open Forum Infect Dis. 2018;5(7):ofy161.
- CDC DPDx: Amebiasis.
- Medscape: Amebiasis Clinical Presentation.
- Medscape: Amebiasis Overview.
- NCBI Bookshelf: Amebiasis.
MSc. Kim Ngoc Son
MSc. Nguyen Thanh Tam

