Health communication
CONTENTS
Cefiderocol: the “trojan horse” warrior in the era of multidrug-resistant bacteria
1. General Introduction
Cefiderocol is an injectable siderophore cephalosporin antibiotic developed by Shionogi & Co., Ltd. (Japan). It is a novel antimicrobial agent specifically designed to address infections caused by multidrug-resistant (MDR) and carbapenem-resistant Gram-negative bacteria, including carbapenemase-producing strains.
The most distinctive feature of cefiderocol is its unique mechanism of entry into bacterial cells, which enables it to overcome resistance barriers that many other antibiotics, including carbapenems and newer-generation cephalosporins, have failed to penetrate.
2. Mechanism of Action: The “Trojan Horse” Strategy
Cefiderocol has a unique hybrid chemical structure, sharing similarities with both ceftazidime (third-generation cephalosporin) and cefepime (fourth-generation cephalosporin), but with an additional catechol side chain at the C-3 position. Its antibacterial activity is based on two principal mechanisms:
a. Siderophore-mediated entry (“Trojan Horse” mechanism)
- The catechol moiety of cefiderocol is capable of chelating free iron in the surrounding environment.
- Gram-negative bacteria require iron for growth and possess active iron transport channels on their cell membranes. By binding to iron, cefiderocol effectively “disguises” itself, causing bacteria to mistakenly recognize and actively transport the drug–iron complex across the outer membrane into the periplasmic space.
- Through this mechanism, cefiderocol achieves high concentrations within the periplasmic compartment and is able to overcome resistance related to loss of porin channels, a common mechanism in drug-resistant bacteria.
b. Inhibition of cell wall synthesis
Once the bacterial cell, cefiderocol dissociates from iron and binds to penicillin-binding proteins (PBPs), primarily PBP3, thereby inhibiting peptidoglycan synthesis and leading to bacterial cell death.
.jpg)
Figure 1. Chemical structure of cefiderocol and the characteristics of its structural components.
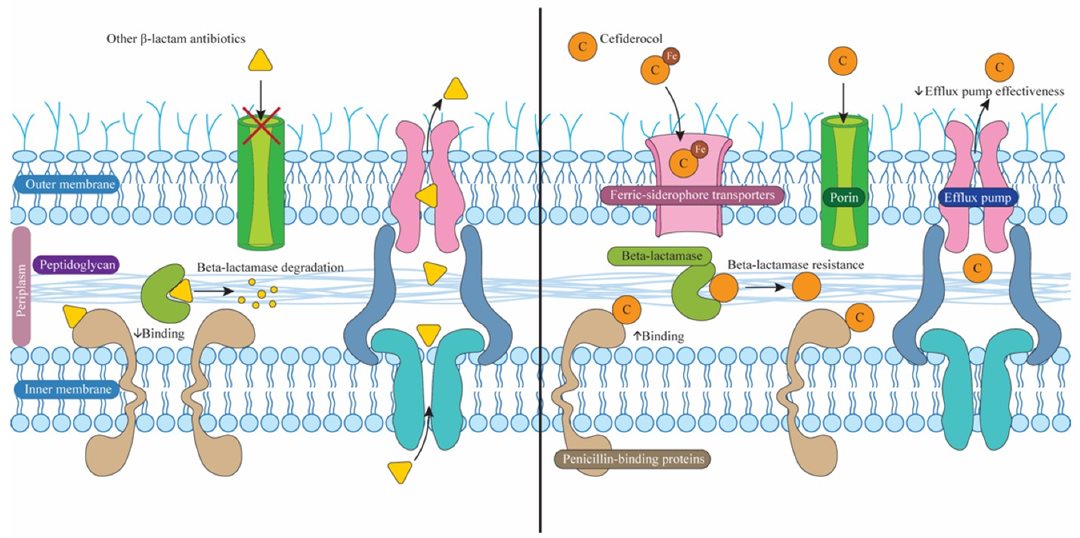
Figure 2. Key differences in antibacterial activity between cefiderocol and other β-lactam antibiotics. Unlike most β-lactams, cefiderocol can enter bacteria via the iron–siderophore transport system and has greater permeability through porin channels. In addition, its increased affinity for penicillin-binding proteins (PBPs), together with reduced susceptibility to efflux pump-mediated extrusion, further enhances its antibacterial efficacy.
3. Antibacterial Spectrum and Advantages/Limitations
Advantages (activity against Gram-negative bacteria)
Cefiderocol demonstrates potent and stable activity against most β-lactamases produced by Gram-negative bacteria:
- Activity against multidrug-resistant organisms: It is highly active against carbapenem-resistant Acinetobacter baumannii, Pseudomonas aeruginosa, and Stenotrophomonas maltophilia, while maintaining a safety and tolerability profile similar to that of other cephalosporins.
- Stability against β-lactamases: Cefiderocol is stable against all four Ambler classes of β-lactamases: Class A (ESBLs, KPC), Class C (AmpC), Class D (OXA), and it is the first agent with activity against Class B β-lactamases. This is a major advantage over newer combinations such as ceftazidime-avibactam and ceftolozane-tazobactam.
Disadvantages and limitations
- Gram-positive bacteria: Cefiderocol has poor or no activity against Gram-positive organisms such as Staphylococcus aureus or Enterococcus faecalis.
- Anaerobic bacteria: It has little or no activity against anaerobic bacteria such as Bacteroides spp. or Clostridioides difficile.
4. Clinical Evidence
In the phase 2 APEKS-cUTI clinical trial comparing cefiderocol with imipenem/cilastatin in patients with complicated urinary tract infection (cUTI):
- Efficacy: Cefiderocol met the criterion for non-inferiority and even demonstrated superiority over imipenem/cilastatin with respect to the composite endpoint of clinical and microbiological response.
- Microbiological eradication rate: The eradication rate was significantly higher with cefiderocol (73%) than with imipenem/cilastatin (56%).
5. Adverse Drug Reactions (ADRs)
Cefiderocol is generally well tolerated and has a safety profile comparable to that of other cephalosporins.
- Common adverse effects: gastrointestinal disturbances (diarrhea, constipation, nausea, abdominal pain) and injection-site reactions (pain, phlebitis).
- Special considerations:
- In clinical trials, the incidence of Clostridioides difficile infection was lower in the cefiderocol group than in the imipenem/cilastatin group.
- Elevated liver enzymes and rash have been reported, but these events are usually mild.
6. Target Population and Place in Therapy
Cefiderocol is positioned as a “reserve weapon” and should be considered for:
- Adult patients with severe infections caused by aerobic Gram-negative bacteria.
- Cases of multidrug-resistant (MDR) or extensively drug-resistant (XDR) infections in which alternative treatment options are limited, particularly infections caused by carbapenem-resistant A. baumannii, P. aeruginosa, or metallo-β-lactamase-producing Enterobacterales.
- Potential indications include complicated urinary tract infections (cUTI), hospital-acquired pneumonia (HAP), ventilator-associated pneumonia (VAP), and bloodstream infections.
In summary, the emergence of cefiderocol represents a major breakthrough in modern medicine, offering a “last ray of hope” for patients with multidrug-resistant and carbapenem-resistant Gram-negative bacterial infections who face limited therapeutic options. With its unique “Trojan Horse” entry mechanism, which enables it to overcome some of the most formidable bacterial resistance barriers, cefiderocol deserves recognition as a strategic weapon in the antimicrobial armamentarium. However, to preserve the long-term effectiveness of this “warrior,” its use must strictly follow antimicrobial stewardship principles, ensuring the right patient, the right indication, and the right dose.
REFERENCES
- Zhanel, G. G., Golden, A. R., Zelenitsky, S., et al. (2019). Cefiderocol: A siderophore cephalosporin with activity against carbapenem-resistant and multidrug-resistant Gram-negative bacilli. Drugs, 79(3), 271–289.
- Ito, A., Sato, T., Ota, M., et al. (2018). In vitro antibacterial properties of cefiderocol, a novel siderophore cephalosporin, against Gram-negative bacteria. Antimicrobial Agents and Chemotherapy, 62(1), e01454-17.
- Portsmouth, S., van Veenhuyzen, D., Echols, R., et al. (2018). Cefiderocol versus imipenem-cilastatin for the treatment of complicated urinary tract infections caused by Gram-negative uropathogens: a phase 2, randomised, double-blind, non-inferiority trial. The Lancet Infectious Diseases, 18(12), 1319–1328.
MSc. Nguyen Yen Ha
MSc. Nguyen Xuan Tu

